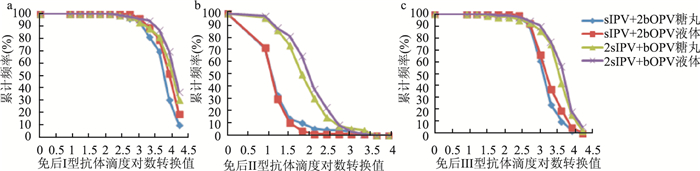
Comparing the antibody titers of sequential program of Sabin strain-based inactivated poliovirus vaccine followed by bivalent types 1 and 3 oral poliovirus vaccine in different dosage forms
-
摘要:
目的 比较Sabin株脊髓灰质炎灭活疫苗(Sabin strain-based inactivated poliovirus vaccine,sIPV)与不同剂型Ⅰ型Ⅲ型脊髓灰质炎减毒活疫苗(bivalent types Ⅰ and Ⅲ oral poliovirus vaccine,bOPV)序贯接种的抗体滴度。 方法 选择柳州400名2月龄婴儿,按1:1:1:1随机分为sIPV+2bOPV糖丸、sIPV+2bOPV液体、2sIPV+bOPV糖丸、2sIPV+bOPV液体4组,按0 d、28 d、56 d免疫程序接种疫苗。检测免前、免后28 d脊灰中和抗体滴度。 结果 序贯程序相同时,1剂sIPV糖丸组与液体组免后Ⅰ型抗体滴度分布差异有统计学意义(Z=-2.589,P=0.010),Ⅱ型(Z=-0.331,P=0.741)和Ⅲ型(Z=-1.556,P=0.120)差异均无统计学意义(均有P>0.05);2剂sIPV糖丸组与液体组免后Ⅰ型(Z=-1.249,P=0.212)、Ⅱ型(Z=-1.658,P=0.097)和Ⅲ型(Z=-1.436,P=0.151)抗体滴度分布差异均无统计学意义(均有P>0.05)。剂型相同时,2剂sIPV组与1剂sIPV组各型抗体滴度分布差异均有统计学意义(均有P < 0.05)(1剂sIPV液体组与2剂sIPV液体组:Ⅰ型:Z=-2.766,P=0.006;Ⅱ型:Z=-9.137,P < 0.001;Ⅲ型:Z=-5.529,P < 0.001。1剂sIPV糖丸组与2剂sIPV糖丸组:Ⅰ型:Z=-3.748,P < 0.001;Ⅱ型:Z=-7.660,P < 0.001;Ⅲ型:Z=-6.030,P < 0.001)。 结论 bOPV糖丸和液体剂型免疫效果相似,各地接种部门应结合疫苗效果、接种对象特征和人口密度,选择适宜剂型接种;如sIPV供应充足,建议优先选择2剂sIPV序贯程序完成脊灰基础免疫接种。 Abstract:Objective To evaluate the antibody titer distributions after primary vaccination by different sequential schedules of Sabin strain-based inactivated poliovirus vaccine(sIPV) and bivalent oral attenuated live poliomyelitis vaccine against types 1 and 3 (bOPV) in Drug Candy(DC) form or liquid dosage form. Methods Eligible infants of 2 months old selected in Liuzhou were assigned randomly in a ratio of 1:1:1:1 to 4 groups as following: sIPV+2bOPV(DC), sIPV+2bOPV(liquid), 2sIPV+bOPV(DC), 2sIPV+bOPV(liquid), and were vaccinated at 0, 28, 56 days. Polio neutralizing antibody titers against poliovirus types 1, 2 and 3 were tested prior to Dose 1 and at 28 days after Dose 3. Results The antibody titer distribution for type 1 was statistically different between sIPV+2bOPV(DC) and sIPV+2bOPV(liquid) (Z=-2.589, P=0.010) while no significant differences were detected between the two groups for type 2(Z=-0.331, P=0.741) and type 3(Z=-1.556, P=0.120). There were no significant differences between 2sIPV +bOPV(DC) and 2sIPV+bOPV(liquid) for the distributions(All P>0.05) (type 1:Z=-1.249, P=0.212; type 2:Z=-1.658, P=0.097; type 3:Z=-1.436, P=0.151). In the same dosage forms with different sequential schedules, the antibody titer distributions were significantly different between 2 doses sIPV and 1 dose sIPV groups(All P < 0.05)(sIPV+2bOPV(liquid) vs 2sIPV+bOPV(liquid): type 1:Z=-2.766, P=0.006; type 2:Z=-9.137, P < 0.001; type 3:Z=-5.529, P < 0.001. sIPV+2bOPV(DC) vs 2sIPV+bOPV(DC): type 1:Z=-3.748, P < 0.001; type 2:Z=-7.660, P < 0.001; type 3:Z=-6.030, P < 0.001). Conclusions Different dosage forms have similar immune effects, so appropriate dosage forms should be selected for vaccination according to the effectiveness, characteristics of subjects and the population density. In the case of sufficient supply of sIPV, 2 doses sIPV sequential program should be the first choice to complete the primary immunization. -
Key words:
- Poliomyelitis /
- Inactivated vaccine /
- Attenuated vaccine /
- Sequential schedule /
- Dosage forms
-
表 1 免后各组Ⅰ型抗体滴度分布比较[n(%)]
Table 1. Comparison of the neutralizing antibody distributions against poliovirus type 1[n(%)]
抗体滴度 sIPV+2bOPV糖丸 sIPV+2bOPV液体 2sIPV+bOPV糖丸 2sIPV+bOPV液体 128~ 0(0.00) 0(0.00) 1(1.14) 1(1.09) 256~ 3(3.45) 0(0.00) 1(1.14) 1(1.09) 512~ 2(2.30) 3(3.61) 4(4.55) 2(2.17) 1 024~ 11(12.64) 6(7.23) 4(4.55) 1(1.09) 2 048~ 10(11.49) 8(9.64) 6(6.82) 7(7.61) 4 096~ 34(39.08) 23(27.71) 18(20.45) 16(17.39) 8 192~ 18(20.69) 27(32.53) 27(30.68) 30(32.61) ≥16 384 9(10.34) 16(19.28) 27(30.68) 34(36.96) 合计 87(100.00) 83(100.00) 88(100.00) 92(100.00) 注:4组间比较:χ2=29.385,P<0.001。组间两两比较:sIPV+2bOPV糖丸vs液体Z=-2.589,P=0.010;2sIPV+bOPV糖丸vs液体Z=-1.249,P=0.212;sIPV+2bOPV糖丸vs 2sIPV+bOPV糖丸Z=-3.748,P<0.001;sIPV+2bOPV液体vs 2sIPV+bOPV液体Z=-2.766,P=0.006。 表 2 免后各组Ⅱ型抗体滴度分布比较[n(%)]
Table 2. Comparison of the neutralizing antibody distributions against poliovirus type 2[n(%)]
抗体滴度 sIPV+2bOPV糖丸 sIPV+2bOPV液体 2sIPV+bOPV糖丸 2sIPV+bOPV液体 0~ 25(28.74) 23(27.71) 3(3.41) 2(2.17) 8~ 33(37.93) 35(42.17) 9(10.23) 9(9.78) 16~ 17(19.54) 16(19.28) 11(12.50) 6(6.52) 32~ 3(3.45) 6(7.23) 21(23.86) 15(16.30) 64~ 4(4.60) 2(2.41) 17(19.32) 25(27.17) 128~ 1(1.15) 0(0.00) 14(15.91) 14(15.22) 256~ 0(0.00) 0(0.00) 6(6.82) 13(14.13) 512~ 1(1.15) 0(0.00) 2(2.27) 5(5.43) 1 024~ 2(2.30) 0(0.00) 1(1.14) 2(2.17) 2 048~4 096 1(1.15) 1(1.20) 4(4.55) 1(1.09) 合计 87(100.00) 83(100.00) 88(100.00) 92(100.00) 注:4组间比较:χ2=142.667,P<0.001。组间两两比较:sIPV+2bOPV糖丸vs液体Z=-0.331,P=0.741;2sIPV+bOPV糖丸vs液体Z=-1.658,P=0.097;sIPV+2bOPV糖丸vs 2sIPV+bOPV糖丸Z=-7.660,P<0.001;sIPV+2bOPV液体vs 2sIPV+bOPV液体Z=-9.137,P<0.001。 表 3 免后各组Ⅲ型抗体滴度分布比较[n(%)]
Table 3. Comparison of the neutralizing antibody distributions against poliovirus type 3[n(%)]
抗体滴度 sIPV+2bOPV糖丸 sIPV+2bOPV液体 2sIPV+bOPV糖丸 2sIPV+bOPV液体 16~ 0(0.00) 0(0.00) 1(1.14) 0(0.00) 32~ 0(0.00) 0(0.00) 1(1.14) 1(1.09) 64~ 1(1.15) 0(0.00) 0(0.00) 0(0.00) 128~ 2(2.30) 1(1.20) 2(2.27) 1(1.09) 256~ 5(5.75) 5(6.02) 2(2.27) 3(3.26) 512~ 26(29.89) 22(26.51) 6(6.82) 4(4.35) 1 024~ 32(36.78) 24(28.92) 10(11.36) 12(13.04) 2 048~ 12(13.79) 15(18.07) 29(32.95) 19(20.65) 4 096~ 7(8.05) 12(14.46) 23(26.14) 35(38.04) 8 192~ 2(2.30) 4(4.82) 12(13.64) 12(13.04) ≥16 384 0(0.00) 0(0.00) 2(2.27) 5(5.43) 合计 87(100.00) 83(100.00) 88(100.00) 92(100.00) 注:4组间比较:χ2=70.453,P<0.001。组间两两比较:sIPV+2bOPV糖丸vs液体Z=-1.556,P=0.120;2sIPV+bOPV糖丸vs液体Z=-1.436,P=0.151;sIPV+2bOPV糖丸vs 2sIPV+bOPV糖丸Z=-6.030,P<0.001;sIPV+2bOPV液体vs 2sIPV+bOPV液体Z=-5.529,P<0.001。 -
[1] 杨婵, 周莉薇, 李吴萍, 等. 银川市儿童监护人脊灰疫苗知晓率及其影响因素分析[J]. 中华疾病控制杂志, 2016, 20(8): 805-808. DOI: 10.16462/j.cnki.zhjbkz.2016.08.013.Yang C, Zhou LW, Li WP, et al. Analysis on the awareness rate of poliomyelitis vaccine and influential factors among guardians of children in Yinchuan City[J]. Chin J Dis Control Prev, 2016, 20(8): 805-808. DOI: 10.16462/j.cnki.zhjbkz.2016.08.013. [2] 熊玮仪, 董铎. 消灭脊灰后期的免疫策略及脊灰疫苗不同免疫程序的安全性比较[J]. 中国药物警戒, 2017, 14(2): 90-94. https://www.cnki.com.cn/Article/CJFDTOTAL-YWJJ201702008.htmXiong WY, Dong D. Review on the immunization strategy in Polio Post-eradication Era and comparison of the safety of polio vaccination programs[J]. Chin J Pharmacovigil, 2017, 14(2): 90-94. https://www.cnki.com.cn/Article/CJFDTOTAL-YWJJ201702008.htm [3] Jorba J, Diop OM, Iber J, et al. Update on vaccine-derived polioviruses-worldwide, January 2016-June 2017[J]. Mmwr Morb Mortal Weekly Rep, 2017, 66(43): 1185-1191. DOI: 10.15585/mmwr.mm6643a6. [4] Liao GY, Li RC, Li CG, et al. Safety and immunogenicity of inactivated poliovirus vaccine made from Sabin strains: a phase Ⅱ, randomized, positive-controlled trial[J]. J Infect Dis, 2012, 205(2): 237-243. DOI: 10.1093/infdis/jir723. [5] Lu L, Li XM, Zhang HR, et al. Immunogenicity and persistence from different 3-dose schedules of live and inactivated polio vaccines in Chinese infants[J]. Vaccine, 2015, 33(36): 4653-4658. DOI: 10.1016/j.vaccine.2014.08.091. [6] 石晓娟, 刘兰, 周莉薇, 等. 国产脊髓灰质炎灭活疫苗和口服减毒活疫苗序贯接种基础免疫效果评价[J]. 中国生物制品学杂志, 2018, 31(5): 521-522. DOI: 10.13200/j.cnki.cjb.002177.Shi XJ, Liu L, Zhou LW, et al. Evaluation on effect of sequential immunization with inactivated poliovirus vaccine and oral live attenuated poliomyelitis vaccine[J]. Chin J Biologicals, 2018, 31(5): 521-522. DOI: 10.13200/j.cnki.cjb.002177. [7] Qiu JJ, Yang YK, Huang LR, et al. Immunogenicity and safety evaluation of bivalent types 1 and 3 oral poliovirus vaccine by comparing different poliomyelitis vaccination schedules in China: A randomized controlled non-inferiority clinical trial[J]. Hum Vaccin Immunother, 2017, 13(6): 1-10. DOI: 10.1080/21645515.2017.1288769. [8] Sutter RW, Bahl S, Deshpande JM, et al. Immunogenicity of a new routine vaccination schedule for global poliomyelitis prevention: an open-label, randomised controlled trial[J]. Lancet, 2015, 386(10011): 2413-2421. DOI: 10.1016/S0140-6736(15)00237-8. [9] O'Ryan M, Bandyopadhyay AS, Villena R. Corrections. Inactivated poliovirus vaccine given alone or in a sequential schedule with bivalent oral poliovirus vaccine in Chilean infants: a randomised, controlled, open-label, phase 4, non-inferiority study[J]. Lancet Infect Dis, 2015, 15(10): 1273-1282. DOI: 10.1016/S1473-3099(15)00219-4. -





 下载:
下载: