The correlation between bisphenol A exposure and ceramide as well as serum tumor markers in colorectal cancer
-
摘要:
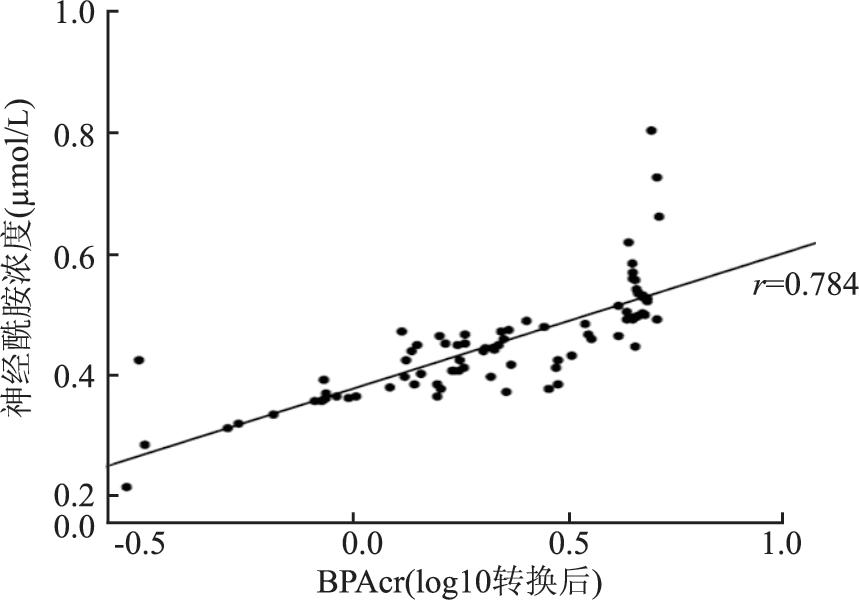
目的 探讨双酚A(bisphenol A, BPA)暴露与结直肠癌(colorectal cancer, CRC)肿瘤组织神经酰胺(ceramide, Cer)及血清肿瘤标志物的相关性。 方法 收集84例CRC患者的晨尿、CRC肿瘤组织, 使用液相色谱-质谱联法(liquid chromatography mass spectrometer, LC-MS)检测尿液BPA浓度, 尿BPA浓度用尿肌酐(creatinine, Cr)进行校正(BPAcr)。酶联免疫吸附(enzyme-linked immunosorbent assay, ELISA)法检测肿瘤组织Cer浓度, 分析尿BPAcr与肿瘤组织Cer、血清肿瘤标志物之间的相关性。 结果 CRC肿瘤组织Cer与BPAcr呈正相关(r=0.784, P < 0.001), 回归分析显示肿瘤组织Cer与BPAcr的回归系数为0.218(95% CI:0.18~0.26), 具有统计学意义(P < 0.001);血清CEA阳性和阴性、CA125阳性和阴性、CA19-9阳性和阴性组之间CRC肿瘤组织Cer和尿BPAcr差异均有统计学意义(均有P < 0.05), 而AFP阳性和阴性组间肿瘤组织Cer和尿BPAcr差异无统计学意义(P=0.247)。血清CEA、CA125和CA19-9水平与尿BPAcr(r值分别为0.348, 0.251, 0.281, 均有P < 0.05)和CRC肿瘤组织Cer(r值分别为0.265, 0.309, 0.263, 均有P < 0.05)均呈正相关。 结论 BPA暴露可能会引起CRC肿瘤组织内Cer增加以及血清肿瘤标志物的异常, 提示BPA暴露可能通过影响CRC肿瘤组织Cer代谢参与CRC的发生发展。 Abstract:Objective The aim is to investigate the correlation between bisphenol A(BPA) exposure and tumor tissue ceramide(Cer) as well as serum tumor markers in colorectal cancer(CRC). Methods The morning urine and CRC tumor tissue were collected from 84 patients with CRC. The concentration of urine BPA was determined by liquid chromatography-mass spectrometer(LC-MS), urine BPA concentration was corrected with creatinine(Cr). Cer concentration of CRC tumor tissue was detected by Enzyme-linked immunosorbent assay(ELISA). The correlations of urine BPAcr, Cer content of CRC tumor tissue and tumor markers were analyzed. Results Cer content in CRC tumor tissue was positively correlated with BPAcr(r=0.784, P < 0.001). Regression analysis showed that the regression coefficient of Cer content in CRC tumor tissue and BPAcr was 0.218(95%CI: 0.18-0.26), which was statistically significant(P < 0.001). There were significantly differences in CRC tumor tissue Cer and urine BPAcr between the CEA positive and negative groups, CA125 positive and negative groups, and CA19-9 positive and negative groups(allP < 0.05), while there was no significant difference between AFP positive and negative groups in CRC tumor tissue Cer and urine BPAcr(P=0.247). Serum CEA, CA125 and CA19-9 were positively correlated with urine BPAcr(r values were 0.348, 0.251, 0.281, respectively, all P < 0.05) and Cer content in CRC tumor tissue(r values were 0.265, 0.309, 0.263, respectively, all P < 0.05). Conclusions BPA exposure may cause an increase of Cer in CRC tumor tissue and abnormalities in serum tumor markers, suggesting that BPA exposure may participate in the development and occurance of CRC by affecting the metabolism of Cer in CRC tumor tissue. -
Key words:
- Bisphenol A /
- Colorectal cancer /
- Ceramide /
- Tumor markers
-
表 1 CRC患者一般情况,尿BPAcr,肿瘤组织Cer和血清肿瘤标志物浓度
Table 1. General characteristics of CRC patients and the concentrations of urine BPAcr, tumor tissue Cer and serum tumor markers
变量 (n=84) 年龄(x±s, 岁) 61.73±10.97 性别 男(n) 46 女(n) 38 BMI (x±s, kg/m2) 23.59±3.33 CEA[M(P25, P75), ng/ml] 18.92(3.09, 20.00) 阳性(n) 56 阴性(n) 28 CA125[M(P25, P75), U/ml] 28.80(16.64, 38.56) 阳性(n) 40 阴性(n) 44 CA19-9[M(P25, P75), U/ml] 37.00(31.00, 43.92) 阳性(n) 44 阴性(n) 40 AFP[M(P25, P75), ng/ml] 3.30(1.98, 8.40) 阳性(n) 29 阴性(n) 55 尿BPAcr[M(P25, P75), μg/g] 2.21(1.38, 4.45) Cer (x±s, μmol/g) 0.45±0.09 表 2 CRC肿瘤组织Cer与尿BPAcr的多重线性回归分析
Table 2. Multiple linear regression analysis of CRC tumor tissue Cer and urine BPAcr
变量 β sx 标准化β t值 P值 95% CI值 常量 0.472 0.060 7.905 < 0.001 0.353~0.591 性别a -0.012 0.013 -0.067 -0.951 0.344 -0.037~0.013 年龄 -0.001 0.001 -0.094 -1.343 0.183 -0.002~0.001 BMI -0.001 0.002 -0.041 -0.592 0.556 -0.005~0.003 尿BPAcr b 0.218 0.020 0.769 10.911 < 0.001 0.178~0.258 注:a表示统计分析时分别将男、女赋值:“男”=“1”, “女”=“2”; b表示尿BPAcr进行log 10转换。 表 3 血清肿瘤标志物与尿BPAcr相关性分析
Table 3. Analysis of correlation between serum tumor markers and urine BPAcr
血清肿瘤标志物 n 相关系数 P值 CEA 84 0.348 0.001 CA125 84 0.251 0.022 CA19-9 84 0.281 0.009 AFP 84 -0.113 0.308 表 4 CRC肿瘤组织Cer与血清肿瘤标志物的相关性分析
Table 4. Correlation analysis between CRC tumor tissue Cer and serum tumor markers
血清肿瘤标志物 n 相关系数 P值 CEA 84 0.265 0.015 CA125 84 0.309 0.004 CA19-9 84 0.263 0.030 AFP 84 -0.050 0.653 -
[1] Hines CJ, Jackson MV, Deddens JA, et al. Urinary bisphenol A(BPA)concentrations among workers in industries that manufacture and use BPA in the USA[J]. Ann Work Expo Health, 2017, 61(2): 164-182. DOI: 10.1093/annweh/wxw021. [2] Le HH, Carlson EM, Chua JP, et al. Bisphenol A is released from polycarbonate drinking bottles and mimics the neurotoxic actions of estrogen in developing cerebellar neurons[J]. Toxicol Lett, 2008, 176(2): 149-156. DOI: 10.1016/j.toxlet.2007.11.001. [3] Chen ZJ, Yang XL, Liu H, et al. Bisphenol A modulates colorectal cancer protein profile and promotes the metastasis via induction of epithelial to mesenchymal transitions[J]. Arch Toxicol, 2015, 89(8): 1371-1381. DOI: 10.1007/s00204-014-1301-z. [4] Rochester JR. Bisphenol A and human health: a review of the literature[J]. Reprod Toxicol, 2013, 42(12): 132-155. DOI: 10.1016/j.reprotox.2013.08.008. [5] Arnold M, Sierra MS, Laversanne M, et al. Global patterns and trends in colorectal cancer incidence and mortality[J]. Gut, 2017, 66(4): 683-691. DOI: 10.1136/gutjnl-2015-310912. [6] Kurz J, Parnham MJ, Geisslinger G, et al. Ceramides as novel disease biomarkers[J]. Trends Mol Med, 2019, 25(1): 20-32. DOI: 10.1016/j.molmed.2018.10.009. [7] Tarapore P, Ying J, Ouyang B, et al. Exposure to bisphenol A correlates with early-onset prostate cancer and promotes centrosome amplification and anchorage-independent growth in vitro[J]. PLoS One, 2014, 9(3): e90332. DOI: 10.1371/journal.pone.0090332. [8] Shafei A, Ramzy MM, Hegazy AI, et al. The molecular mechanisms of action of the endocrine disrupting chemical bisphenol A in the development of cancer[J]. Gene, 2018, 647: 235-243. DOI: 10.1016/j.gene.2018.01.016. [9] Jasperson KW, Tuohy TM, Neklason DW, et al. Hereditary and familial colon cancer[J]. Gastroenterology, 2010, 138(6): 2044-2058. DOI: 10.1155/1997/859106. [10] 张鑫东, 葛晓蕾, 刘省存, 等.血清CA199和CEA对结直肠癌转移和预后预测的价值[J].中华疾病控制杂志, 2018, 22(1): 57-61. DOI: 10.16462/j.cnki.zhjbkz.2018.01.013.Zhang XD, Ge XL, Liu SC, et al. The prognostic and predictive value of serum CA199 and CEA in colorectal cancer metastasis[J]. Chin J Dis Control Prev, 2018, 22(1): 57-61. DOI: 10.16462/j.cnki.zhjbkz.2018.01.013. [11] Perttula K, Schiffman C, Edmands WMB, et al. Untargeted lipidomic features associated with colorectal cancer in a prospective cohort[J]. BMC Cancer, 2018, 18(1): 996. DOI: 10.1186/s12885-018-4894-4. [12] Ogretmen B, Hannun YA. Biologically active sphingolipids in cancer pathogenesis and treatment[J]. Nat Rev Cancer, 2004, 4(8): 604-616. DOI: 10.1038/nrc1411. [13] Chen L, Chen H, Li Y, et al. Endocannabinoid and ceramide levels are altered in patients with colorectal cancer[J]. Oncol Rep, 2015, 34(1): 447-454. DOI: 10.3892/or.2015.3973. [14] Fitzgerald S, Sheehan KM, Espina V, et al. High CerS5 expression levels associate with reduced patient survival and transition from apoptotic to autophagy signalling pathways in colorectal cancer[J]. J Pathol Clin Res, 2015, 1(1): 54-65. DOI: 10.1002/cjp2.5. -





 下载:
下载: