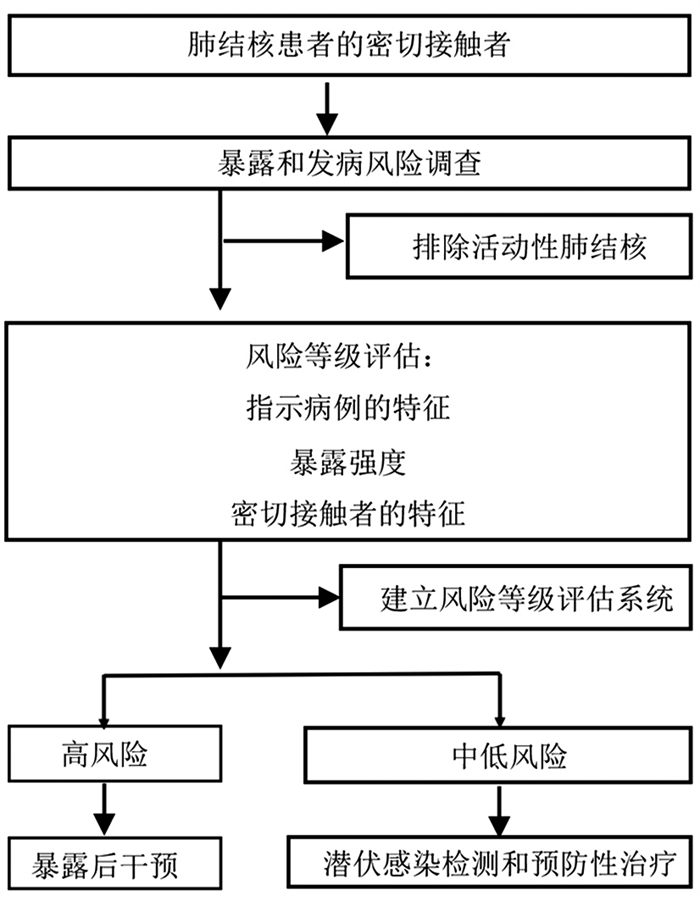
-
摘要: 我国是结核病高负担国家,要实现终结结核病流行的全球战略目标,还需要进一步完善现有的技术体系和防控策略。暴露后干预(post-exposure prophylaxis, PEP)作为一种有效的预防手段在传染病控制工作中得到广泛应用,但是在结核病领域应用较少。目前,与病原学阳性肺结核患者有密切接触的<5岁儿童是被建议开展PEP的唯一群体,主要的干预手段是基于抗结核药物的预防性治疗。本文旨在探讨PEP在结核病预防工作中潜在的应用价值及面临的挑战,为完善肺结核密切接触者的管理工作以及实践结核病防控工作从预防发病到预防感染的“关口前移”提供新的思路。Abstract: China is one of the high burden countries on tuberculosis, current tuberculosis control strategy need to be further improved in order to achieve the End Tuberculosis Strategy. As a widely used effective tool in preventing infectious diseases, post-exposure prophylaxis (PEP) has not yet been systematically applied in tuberculosis control. Currently, children aged < 5 years with close contacts to microbiologically confirmed tuberculosis patients was the only target population recommended to initiate PEP with anti-tuberculosis agents. The present study illustrated the application of PEP in preventing infectious diseases. The values of PEP in tuberculosis prevention and challenge faced were further discussed. It might provide new insight for improving close contacts management and moving tuberculosis control action forward from preventing disease occurrence to controlling infection.
-
Key words:
- Tuberculosis /
- Infectious disease /
- Risk evaluation /
- Post-exposure prophylaxis
-
表 1 暴露后干预和潜伏感染预防性治疗的区别和联系
Table 1. Difference and relationship between post-exposure prophylaxis and preventive treatment of latent infection
项目 密切接触者的暴露后干预 潜伏感染人群的预防性治疗 干预目的 降低有近期暴露史的高风险人群的感染风险和近期感染后的发病风险 降低潜伏感染者发病风险 目标人群 有近期暴露史、感染状态未知的高风险人群,目前国际指南建议的干预对象主要是与活动性肺结核患者有密切接触的<5岁儿童 已明确感染状态且有较高发病风险的潜伏感染者,包括合并HIV感染者、近期感染者、免疫抑制剂使用者、器官移植人群、硅沉着病患者等 干预手段 暴露后化疗、疫苗(预防感染)、生物制剂等 预防性化疗、疫苗(预防发病)、生物制剂等 -
[1] 高磊, 张慧, 胡茂桂, 等. 基于多中心调查数据和空间统计模型的全国结核分枝杆菌潜伏感染率估算[J]. 中国防痨杂志, 2022, 44(1): 54-59. DOI: 10.19982/j.issn.1000-6621.20210661.Gao L, Zhang H, Hu MG, et al. Estimation of the national burden on latent tuberculosis infection based a multi-center epidemiological survey and the space statistics model[J]. Chin J Antituberc, 2022, 44(1): 54-59. DOI: 10.19982/j.issn.1000-6621.20210661 [2] Behr MA, Edelstein PH, Ramakrishnan L. Revisiting the timetable of tuberculosis[J]. BMJ, 2018, 362: k2738. DOI: 10.1136/bmj.k2738. [3] Houben RM, Dodd PJ. The global burden of latent tuberculosis infection: a re-estimation using mathematical modelling[J]. PLoS Med, 2016, 13(10): e1002152. DOI: 10.1371/journal.pmed.1002152. [4] World Health Organization. WHO consolidated guidelines on tuberculosis: tuberculosis preventive treatment: module 1: prevention (2020)[EB/OL]. (2020-03-24)[2021-11-15]. https://apps.who.int/iris/handle/10665/331170. [5] Bader MS, McKinsey DS. Postexposure prophylaxis for common infectious diseases[J]. Am Fam Physician, 2013, 88(1): 25-32. [6] Anibarro L, Trigo M, Villaverde C, et al. Interferon-γ release assays in tuberculosis contacts: is there a window period?[J]. Eur Respir J, 2011, 37(1): 215-217. DOI: 10.1183/09031936.00030610. [7] Malik AA, Farooq S, Jaswal M, et al. Safety and feasibility of 1 month of daily rifapentine plus isoniazid to prevent tuberculosis in children and adolescents: a prospective cohort study[J]. Lancet Child Adolesc Health, 2021, 5(5): 350-356. DOI: 10.1016/S2352-4642(21)00052-3. [8] Martinez L, Cords O, Horsburgh CR, et al. The risk of tuberculosis in children after close exposure: a systematic review and individual-participant meta-analysis[J]. Lancet, 2020, 395(10228): 973-984. DOI: 10.1016/S0140-6736(20)30166-5. [9] Getahun H, Matteelli A, Chaisson RE, et al. Latent Mycobacterium tuberculosis infection[J]. N Engl J Med, 2015, 372(22): 2127-2135. DOI: 10.1056/NEJMra1405427. [10] Saunders MJ, Wingfield T, Datta S, et al. A household-level score to predict the risk of tuberculosis among contacts of patients with tuberculosis: a derivation and external validation prospective cohort study[J]. Lancet Infect Dis, 2020, 20(1): 110-122. DOI: 10.1016/S1473-3099(19)30423-2. [11] Selwyn PA, Hartel D, Lewis VA, et al. A prospective study of the risk of tuberculosis among intravenous drug users with human immunodeficiency virus infection[J]. N Engl J Med, 1989, 320(9): 545-550. DOI: 10.1056/NEJM198903023200901. [12] Saunders MJ, Wingfield T, Tovar MA, et al. A score to predict and stratify risk of tuberculosis in adult contacts of tuberculosis index cases: a prospective derivation and external validation cohort study[J]. Lancet Infect Dis, 2017, 17(11): 1190-1199. DOI: 10.1016/S1473-3099(17)30447-4. [13] World Health Organization. Global tuberculosis report 2020[EB/OL]. (2020-10-15)[2021-11-15]. https://www.who.int/publications/i/item/9789240013131. [14] Stanford J, Stanford C, Grange J. Immunotherapy with Mycobacterium vaccae in the treatment of tuberculosis[J]. Front Biosci, 2004, 9: 1701-1719. DOI: 10.2741/1292. [15] Tang P, Chen X, Xu J, et al. Autologous cytokine-induced killer cell immunotherapy enhances chemotherapy efficacy against multidrug-resistant tuberculosis[J]. J Immunol Res, 2022, 2022: 2943113. DOI: 10.1155/2022/2943113. [16] Matteucci KC, Correa AAS, Costa DL. Recent advances in host-directed therapies for tuberculosis and malaria[J]. Front Cell Infect Microbiol, 2022, 12: 905278. DOI: 10.3389/fcimb.2022.905278. -





 下载:
下载: